
These are the cathode rays.Įnough of the air has been removed from the tube that most of the electrons can travel the length of the tube without striking a gas molecule. When they strike it, they knock large numbers of electrons out of the surface of the metal, which in turn are repelled by the cathode and attracted to the anode or positive electrode. All the positive ions are attracted to the cathode or negative electrode. 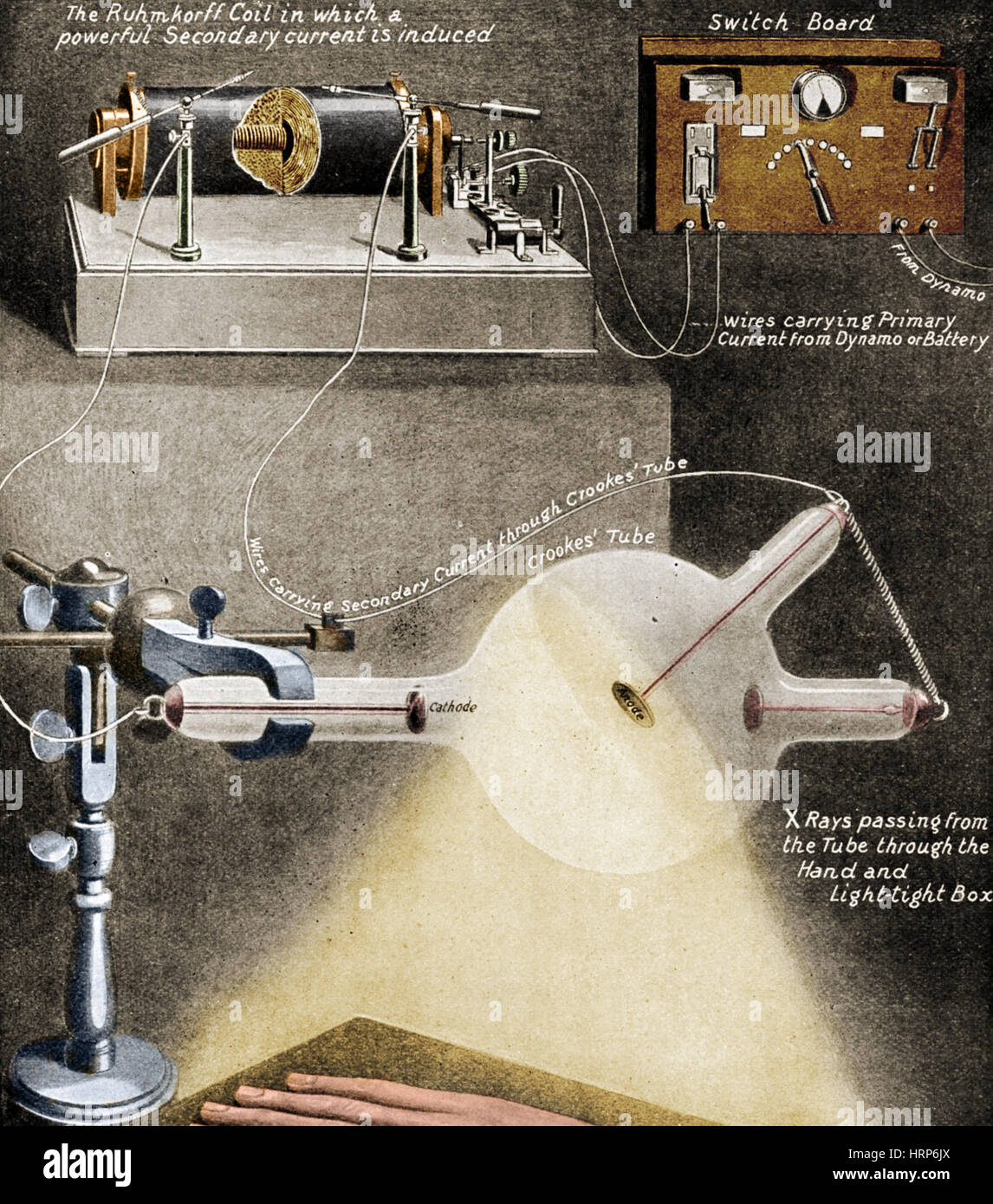
The electrons go on to create more ions and electrons in a chain reaction called a Townsend discharge. The electrons collide with other gas molecules, knocking electrons off them and creating more positive ions. When high voltage is applied to the tube, the electric field accelerates the small number of electrically charged ions and free electrons always present in the gas, created by natural processes like photoionization and radioactivity. The Crookes tubes require a small amount of air in them to function, from about 10 −6 to 5×10 −8 atmosphere (7×10 −4 - 4×10 −5 torr or 0.1-0.006 pascal). Instead, electrons are generated by the ionization of the residual air by a high DC voltage (from a few kilovolts to about 100 kilovolts) applied between the cathode and anode electrodes in the tube, usually by an induction coil (a "Ruhmkorff coil"). The term Crookes tube is also used for the first generation, cold cathode X-ray tubes, which evolved from the experimental Crookes tubes and were used until about 1920.Ĭrookes tubes are cold cathode tubes, meaning that they do not have a heated filament in them that releases electrons as the later electronic vacuum tubes usually do. Wilhelm Röntgen discovered X-rays using the Crookes tube in 1895. Crookes tubes are now used only for demonstrating cathode rays. Thomson's 1897 identification of cathode rays as negatively charged particles, which were later named electrons. It was used by Crookes, Johann Hittorf, Julius Plücker, Eugen Goldstein, Heinrich Hertz, Philipp Lenard, Kristian Birkeland and others to discover the properties of cathode rays, culminating in J.J. When a high voltage is applied between the electrodes, cathode rays ( electrons) are projected in straight lines from the cathode. ĭeveloped from the earlier Geissler tube, the Crookes tube consists of a partially evacuated glass bulb of various shapes, with two metal electrodes, the cathode and the anode, one at either end. The anode is the electrode at the bottom.Ī Crookes tube (also Crookes–Hittorf tube) is an early experimental electrical discharge tube, with partial vacuum, invented by English physicist William Crookes and others around 1869-1875, in which cathode rays, streams of electrons, were discovered. Electrons (cathode rays) travel in straight lines from the cathode (left), as shown by the shadow cast by the metal Maltese cross on the fluorescence of the righthand glass wall of the tube. The figure below from the 1914 catalog of the Otto Pressler company is so similar to the example in the collection that it is hard not to conclude that our tube was manufactured by Pressler.A Crookes tube: light and dark. 142 2014).Įach paddle in our tube has fluorescent strips that glow when struck by electrons-cool. Humphrey’s “A Century-Old Question: Does a Crookes Paddle Wheel Cathode Ray Tube Demonstrate that Electrons Carry Momentum?” (The Physics Teacher Vol. For more information about this topic, please check out T.E. Thompson, the turning of the wheel was due to the radiometric effect, i.e., the heating of the gas molecules adjacent to the area of the paddles being struck by the cathode rays. As explained by the “discoverer” of the electron, J.J. While the tube’s cathode rays (electrons) do possess momentum, the latter is not sufficient to turn the paddles. This movement was originally believed to demonstrate that the cathode rays possessed momentum that they transferred to the paddles. When high voltage is applied across the tube, the paddle wheel travels along the glass "railway track" from the cathode towards the anode. 
This type of gas discharge tube (aka paddle wheel tube) was invented in the 1880s by William Crookes as part of his investigations into the nature of cathode rays (electrons).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
